Case Study: Telecentric Measurement System for Medical Devices—Cardiac Occluders
I. Measurement Background
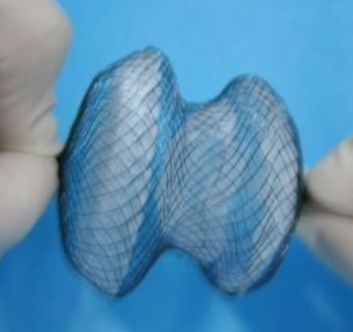
Cardiac occluders are key implantable devices used in interventional cardiology to treat congenital heart defects (such as atrial septal defects, ventricular septal defects, and patent ductus arteriosus) and to occlude the left atrial appendage. They are typically constructed by suturing a highly elastic nickel-titanium alloy scaffold to a occluding membrane and are intended to remain in the body permanently after implantation.
Because cardiac occluders come into direct contact with blood and operate continuously within the beating heart, their dimensional accuracy directly determines the success or failure of the procedure:
-
If the device is too large: It cannot be smoothly inserted into the delivery sheath, or it may exert excessive pressure on cardiac tissue after deployment, leading to pericardial effusion.
-
If the device is too small: It cannot be securely anchored at the defect site, posing a risk of dislodgement and resulting in surgical failure.
Therefore, during the factory quality inspection process, full-dimension measurements of the plug’s waist height, disc diameter, stem length, and contour curvature must be performed with micron-level precision.
II. Measurement Challenges
The unique structure and materials of cardiac occluders pose significant challenges for traditional contact-based measurement methods:
1. High elasticity and susceptibility to deformation: The nickel-titanium alloy mesh is extremely flexible; contact gauges (such as calipers and micrometers) cause the workpiece to undergo elastic deformation upon contact, resulting in severely distorted measurement data that fails to reflect the true dimensions of the workpiece in its free state.
2. Complex structure and obstructions: The occluder has a double-disk mushroom shape, woven from dozens of fine wires, with a surface featuring numerous open mesh patterns. Conventional coordinate measuring machines (CMMs) are limited by depth of field and lighting conditions, making it difficult to clearly capture all edge features.
3. Conflict between efficiency and 100% inspection requirements: As a Class III high-risk medical device, the occluder demands extremely high quality consistency and requires 100% batch inspection. Traditional measurement methods are slow and cannot keep pace with production line rhythms.
III. Solution for Measuring Medical Devices: Cardiac Occluders: HM-1040 Telecentric Measurement System

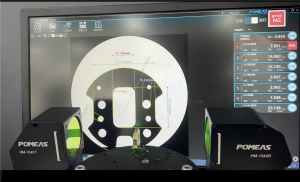
1. Non-contact optical imaging: The HM-1040 captures the edge profile of the occluder instantly using high-definition optical components without coming into contact with the workpiece surface. It can produce clear images even of extremely fine nickel-titanium wires (approximately 0.1–0.3 mm), completely eliminating errors caused by elastic deformation due to contact force.
2. Intelligent Image Processing Algorithms: The instrument’s built-in measurement software automatically identifies and filters out background interference from the mesh frame’s openwork, accurately capturing the virtual envelope diameter, waist height, and coaxiality of the occluder’s outermost edge.
3. Measurement Data and Performance: With a measurement accuracy of 0.001 mm and a repeatability of 1 μm, the system ensures data consistency and stability during mass production.
IV. Measurement Results
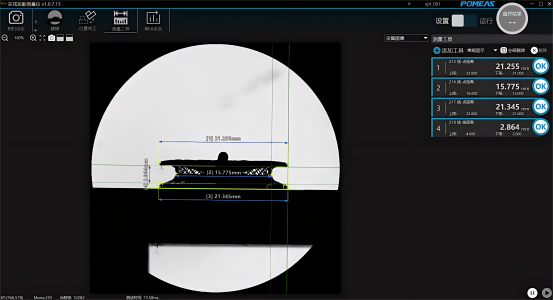

Product recommendation
TECHNICAL SOLUTION
MORE+You may also be interested in the following information
FREE CONSULTING SERVICE
Let’s help you to find the right solution for your project!


 ASK POMEAS
ASK POMEAS  PRICE INQUIRY
PRICE INQUIRY  REQUEST DEMO/TEST
REQUEST DEMO/TEST  FREE TRIAL UNIT
FREE TRIAL UNIT  ACCURATE SELECTION
ACCURATE SELECTION  ADDRESS
ADDRESS Tel:+ 86-0769-2266 0867
Tel:+ 86-0769-2266 0867 Fax:+ 86-0769-2266 0867
Fax:+ 86-0769-2266 0867 E-mail:marketing@pomeas.com
E-mail:marketing@pomeas.com
